Our interdisciplinary lab is interested in clinically-inspired basic and translational research at the intersection between bioengineering and biomedicine. We aim to understand how biophysical and biochemical properties of native extracellular matrix and synthetic biomaterials guide cell response in tissue regeneration, cancer dormancy and bone metastasis. We use materials science and engineering systems, such as scaffolds, hydrogels or microfluidic chips, which obtain inspiration from in vivo imaging and ex vivo multimodal characterization at different length scales.
BIOMATERIALS FOR TISSUE REGENERATION AND DISEASE MODELING


Biomaterial scaffolds and hydrogels allow independent control of biochemical and biophysical properties and have contributed to the understanding of how cells sense these cues in tissue regeneration. We investigate biomaterial physical properties, such as stiffness, 3D architecture or degradation properties, on cell response and tissue regeneration. Such biomaterial systems are also used as platform for disease modeling.
ENGINEERING THE CELL MICROENVIRONMENT
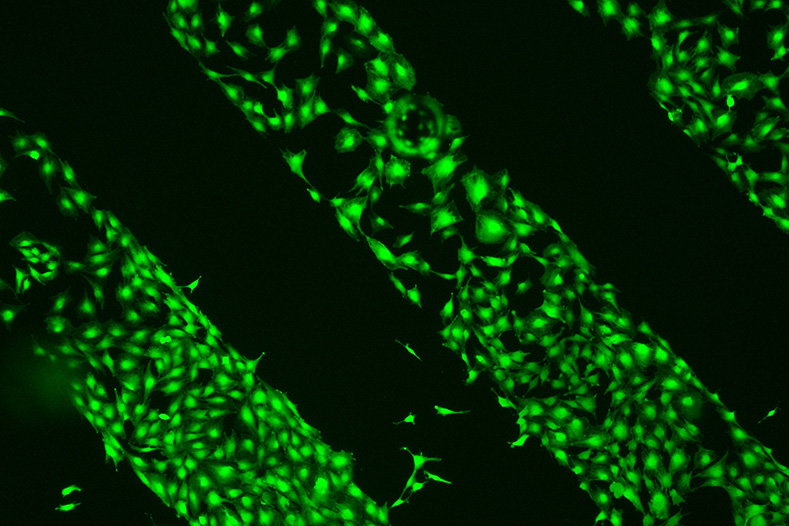
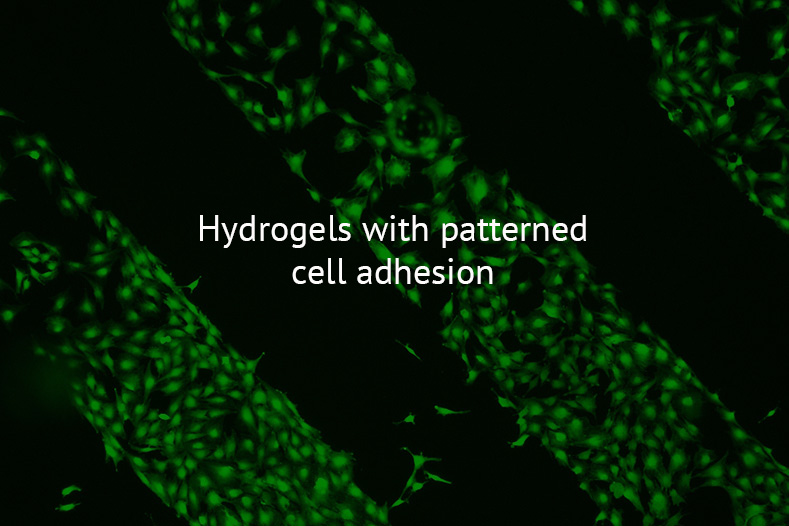
The cell microenvironment or cell niche includes the extracellular matrix, biochemical factors and neighbor cells, and the interaction between them regulates cell function in healing processes as well as in pathological alterations. We use hydrogels to mimic such microenvironments and allow spatial control of hydrogel stiffness, degradability and presentation of polymer-bound biomolecules.
CANCER DORMANCY AND BONE METASTASIS
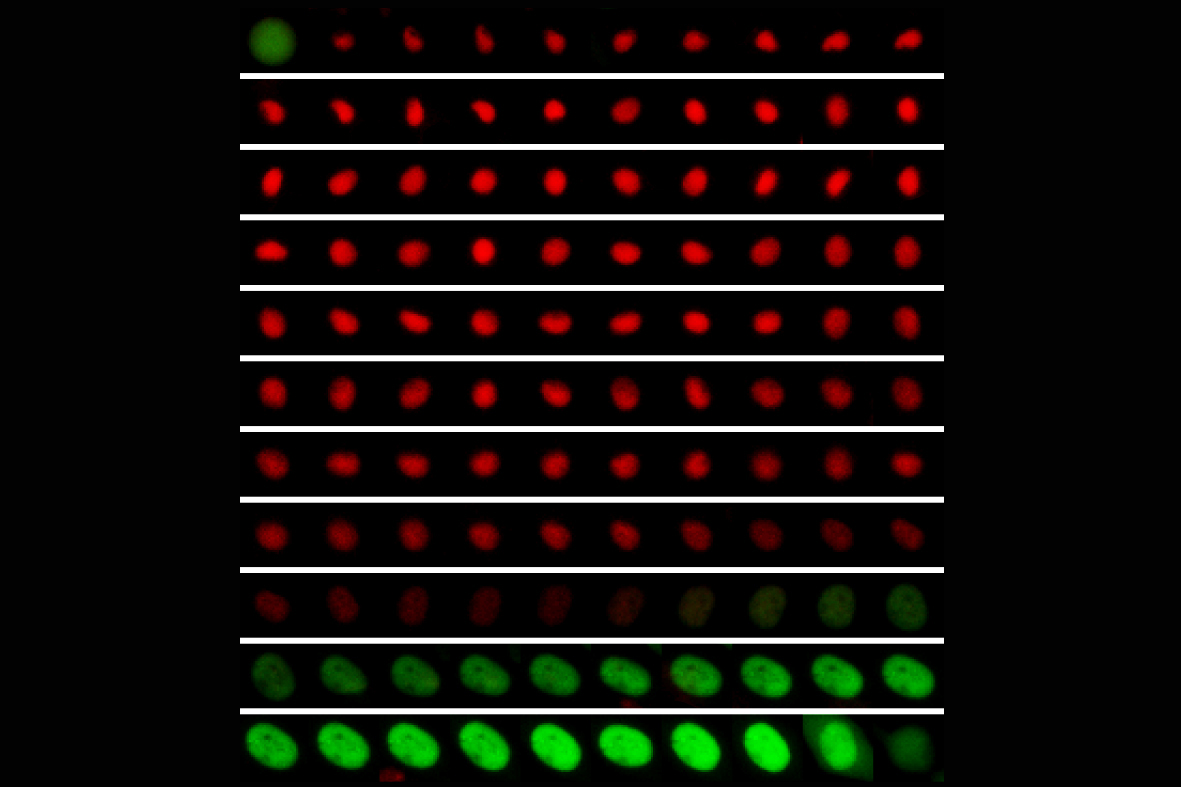

Materials science approaches are also used to investigate cancer progression, such as tumor growth, homing or metastasis. Breast cancer is one of the leading causes of cancer-associated deaths among women worldwide. Breast cancer cells often metastasize to bone. However, metastatic outgrowth might not occur instantly, with time periods for cancer recurrence ranging from years to decades. This indicates that some cells can survive dormant for years before becoming proliferative and leading to metastasis. We aim to elucidate the role of biophysical mechanisms in controlling breast cancer dormancy in the bone marrow, and reactivation leading to bone metastasis. To do so we synthesize biomimetic cell microenvironments and monitor in real time cell cycle dynamics using genetically modified cell lines with FUCCI cell cycle reporter. Such synthetic cell microenvironments obtain inspiration from in vivo models of breast cancer bone metastasis, where we investigate the early metastatic niche by in vivo imaging and ex vivo correlative multiscale characterization.
BIOENGINEERING IN CLINICAL ONCOLOGY
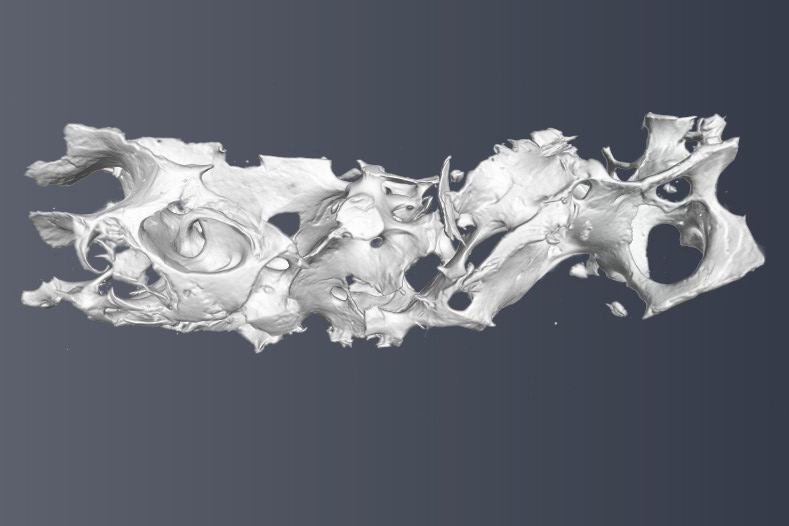
We have strong ties with clinical partners and oncologists at the Charité University Hospital Berlin and at the University Hospital Würzburg, which allows us to address clinically-inspired research questions. In particular, we investigate aberrant extracellular matrix architecture and mechanics in multiple myeloma bone disease and in pancreatic cancer.


